A Complete Guide to Corrective and Preventive Action (CAPA)
Learn the importance of corrective and preventive action for implementing effective quality management systems.

Learn the importance of corrective and preventive action for implementing effective quality management systems.

Published 27 Apr 2026
Article by
7 min read
Corrective and preventive action (CAPA) is a crucial process used to identify and resolve defects and other quality issues in products and processes. Corrective actions eliminate the root cause of existing nonconformances or undesirable situations, while preventive actions are meant to address potential problems before they occur.
CAPA plays a critical role in strengthening a quality management system (QMS) by ensuring problems are addressed at their root rather than at the surface. This structured approach helps organizations resolve nonconformities effectively while reducing the risk of recurring issues.
It also reinforces regulatory compliance and promotes a culture of continuous improvement. By verifying the effectiveness of corrective or preventive actions, organizations improve their process stability, enhance product quality, and maintain accountability across the system.
Implementing an effective CAPA process can have numerous benefits for organizations, including:
Lower operational costs: Reduces rework, scrap, recalls, and downtime.This significantly decreases the overall cost of production for optimized resource allocation across operations.
Stronger regulatory compliance: CAPA supports adherence to standards such as ISO-based systems, making audits smoother and reducing the risk of penalties.
Greater operational efficiency: Streamlines workflows, minimizes waste, and promotes continuous improvement across departments.
Higher customer satisfaction: As issues are addressed at the root, organizations can deliver more dependable products and services, resulting in fewer complaints and stronger customer trust.
Enhanced risk and safety management: A proactive CAPA system identifies potential hazards early and implements preventive measures to reduce incidents and disruptions that could affect business operations.
Data-driven continuous improvement: Frequent monitoring of quality performance using digital tools like checklists provides robust data that can be used for improvement. This provides data-driven performance tracking to refine processes and ensure corrective and preventive actions remain effective over time.
Build from scratch or choose from our collection of free, ready-to-download, and customizable templates.
While CAPA is most commonly used in manufacturing or production, it’s a useful tool that can improve operational tasks across industries, as evaluated in management reviews. Here are practical examples of CAPA in action across different scenarios:
Fixing or replacing defective products found during inspection.
Updating a work procedure after discovering the cause of an error.
Repairing equipment that caused a production issue.
Providing additional training when mistakes are a result of insufficient knowledge.
Monitoring processes regularly to spot possible risks before they escalate.
Scheduling routine equipment maintenance to avoid breakdowns.
Standardizing instructions so everyone follows the same steps.
Reviewing past data trends to gain a better understanding of potential risks and hazards that could affect productivity or performance.
The CAPA process involves various roles and responsibilities to ensure its successful implementation. Here are the key roles and their respective responsibilities in the CAPA process:
CAPA owner: Manages the entire CAPA process, advances actions, aligns cross-functional teams, and ensures compliance with timelines and documentation standards.
Investigator: Conducts root cause analysis by gathering data, reviewing records, and applying structured tools to determine why the issue occurred.
Quality assurance (QA) reviewer: Ensures the CAPA aligns with QMS standards and regulatory requirements by verifying evidence, documentation accuracy, and risk-based actions.
CAPA approver: Reviews and formally approves investigation findings, corrective actions, and closure to confirm that solutions are justified and effective.
Subject Matter Experts (SMEs): Provide specialized technical or process knowledge to validate root causes and ensure corrective and preventive actions are practical and sound.
CAPA board or review committee: Monitors overall CAPA performance, reviews trends, escalations, and overdue cases, and ensures consistent, cross-functional decision-making.
Despite the benefits of implementing a robust CAPA process, organizations may face common challenges. These can include:
CAPA requires employees to understand root cause analysis, documentation standards, and regulatory expectations, which often demands upfront training investment. Without adequate training, investigations become weak, actions lack depth, and the overall system loses effectiveness.
A fragmented or poorly integrated quality framework makes it difficult for CAPA to function effectively. Without a solid system connecting design, production, and quality processes, issues may be addressed in isolation rather than as part of a broader improvement strategy.
It’s important to have the right platform or tools to consistently support smooth implementation of CAPA. Business leaders can opt to use a digital platform that can seamlessly gather and analyze data from teams to ensure all areas are covered.
Cultivate a culture of excellence with our digital solutions that enhance efficiency, agility, and continuous improvement across all operations.
Effective CAPA implementation requires time, personnel, and structured processes, which can strain limited resources. Understaffed or overburdened teams may rush investigations and fail to follow through on corrective actions.
Incomplete or poorly maintained records make it difficult to track investigations, verify effectiveness, and demonstrate compliance. Weak documentation can compromise audits, obscure root causes, and reduce confidence in the CAPA process.
Employees may hesitate to adopt new procedures due to comfort with existing practices or concerns about added workload. Without clear communication and leadership support, resistance can slow adoption and weaken the impact of CAPA initiatives.
To effectively implement a CAPA process and mitigate its common challenges, organizations can follow these key steps:
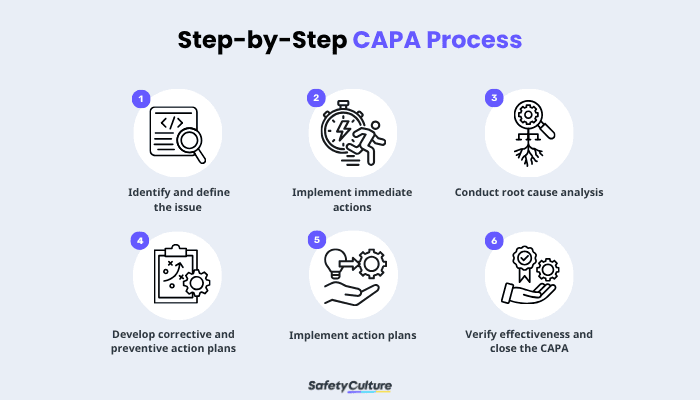
Every CAPA starts by identifying a problem, risk, or nonconformance through complaints, audits, deviations, or trend analysis. The goal is to document clear facts—what happened, when, where, and under what conditions—without assumptions or blame.
Key elements to capture:
Type of issue (e.g., deviation, complaint, audit finding)
Date, location, and process involved
Initial evidence or records
Preliminary risk or impact assessment
Stabilize the situation first to limit additional impact before conducting a deeper investigation. Containment may include stopping a process, isolating affected materials, notifying stakeholders, or applying temporary controls.
This step protects product quality and safety while showing a timely response. Containment is not the final fix but a short-term safeguard while root cause analysis is underway.
Root Cause Analysis (RCA) determines why the issue occurred and how it affected the system in place. The focus of this step should be on process failures rather than individual blame.
Common tools and methods include:
5 Whys for drilling down into cause-and-effect relationships
Ishikawa diagram to explore categories such as Machine, Methods, Materials, People, Measurement, and Environment
FMEA (Failure Mode and Effects Analysis) for structured risk evaluation
Once the root cause is identified, define the specific actions needed to eliminate issues and prevent recurrence. Corrective actions address the identified problem, while preventive actions reduce the likelihood of similar issues happening elsewhere.
Action plans should:
Be directly linked to the root cause
Include assigned responsibilities and deadlines
Be proportionate to the level of risk
Responsible teams carry out approved actions in line with the established plan. This phase often requires coordination across departments such as QA, operations, engineering, or supply chain.
Documentation is critical at this stage to provide clear evidence that teams completed the action plans as intended. Implementation must demonstrate real operational change, not just administrative updates.
After implementation, effectiveness checks confirm that the corrective and preventive actions achieved the desired results. It may involve performance monitoring, audits, trend analysis, or additional data collection over a defined period.
Clear documentation ensures verifiable proof that teams carried out the planned actions. Proper closure demonstrates organizational learning and reinforces a culture of continuous improvement.
SafetyCulture is a mobile-first operations platform adopted across industries such as manufacturing, mining, construction, retail, and hospitality. It’s designed to equip leaders and working teams with the knowledge and tools to do their best work—to the safest and highest standard.
Streamline processes, eliminate bottlenecks, enhance resource utilization, and build an agile and scalable infrastructure with SafetyCulture. Strive for operational excellence to boost competitive advantage, foster sustainable growth, and deliver long-term value.
Save time and reduce costs
Stay on top of risks and incidents
Boost productivity and efficiency
Enhance communication and collaboration
Discover improvement opportunities
Make data-driven business decisions
In this article