What is MDSAP and Why is it Important?
The MDSAP is a certification program for the QMS of manufacturers that wish to sell medical devices to the following MDSAP participating countries: the USA, Australia, Japan, Brazil, and Canada. With that, the Regulatory Authorities (RAs) participating in this program are the following:
Australian Therapeutic Goods Administration (TGA)
Brazil’s Agência Nacional de Vigilância Sanitária (ANVISA)
Health Canada
Ministry of Health, Labour and Welfare (MHLW)/Pharmaceuticals and Medical Devices Agency (PMDA) (Japan)
US FDA
Initiated by the International Medical Device Regulators Forum (IMDRF), MDSAP intends to create a consolidated process for maintaining the safety and quality of medical devices by manufacturers that meet QMS international standards (like ISO 9001 and ISO 13485:2016) and comply with the requirements of Regulatory Authorities (RA) of participating countries.
How to Prepare for MDSAP Certification
To guide you as you prepare for acquiring this certification, here are some steps and tips you can follow:
Gain an understanding of the ISO 13485:2016 standard and the RA requirements of the country where you are to sell medical devices.
Identify areas for improvement in the current QMS by performing a gap analysis or a readiness audit to ensure adherence to ISO 13485:2016 and other requirements.
Conduct quality monitoring audits and maintain a record of results.
Define your organization’s competencies and determine training requirements for MDSAP certification based on the audit results.
Ensure competency needs are met and that all parties involved are kept in the loop.
Partner with an Auditing Organization for MDSAP certification.
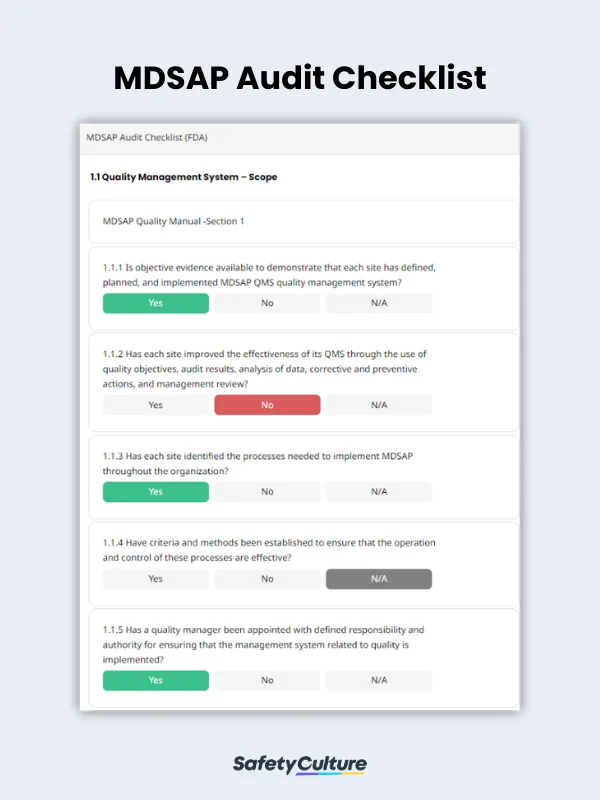
What to Include in an MDSAP Audit Checklist
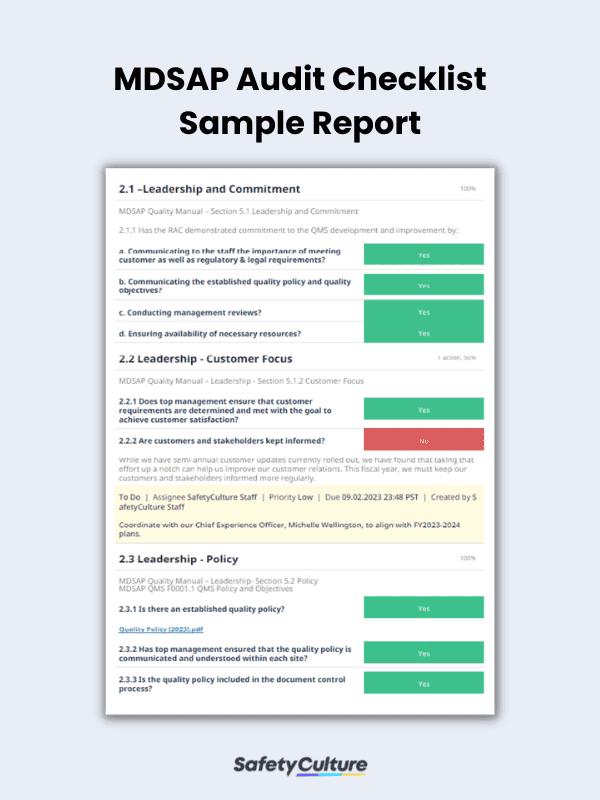
Sample MDSAP Audit Checklist PDF Report | SafetyCulture
To help you conduct an in-depth QMS audit according to MDSAP requirements, use the following guide on what you must put in your MDSAP audit checklist:
Title Page – includes the manufacturing site name, date and location of the audit, and name of the auditor
Individual Sections for Each QMS Requirement – allows you to verify if the manufacturing site follows the standards stated in the MDSAP quality manual
Completion Page – where you can provide comments and recommendations, if any, and sign off the report by including the name and signature of the auditor