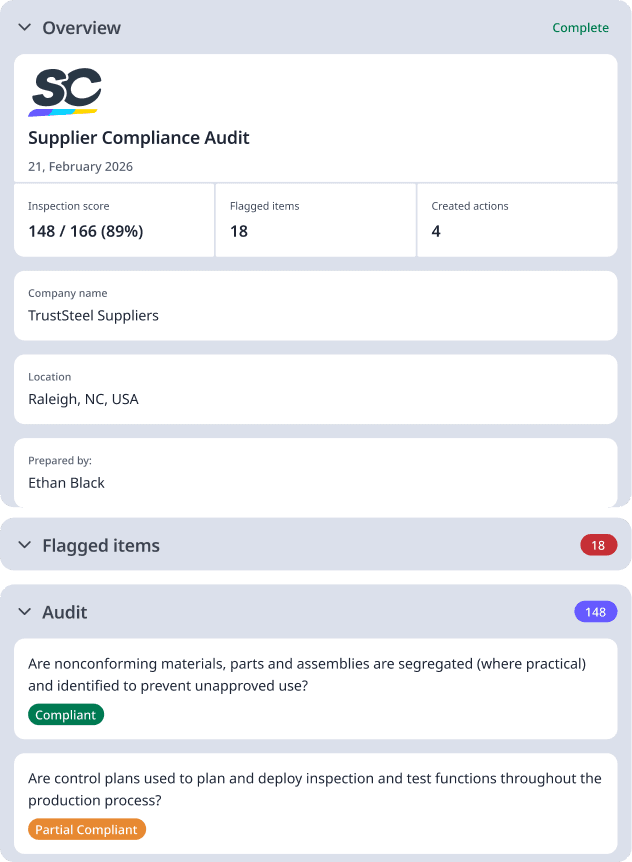
What Is a Supplier Compliance Audit?
A Supplier Compliance Audit is a systematic evaluation of a supplier's ability to consistently meet quality, regulatory, and contractual requirements. These audits assess the supplier's manufacturing processes, quality management systems, documentation practices, and compliance with applicable standards and regulations. They are critical for verifying that suppliers can reliably provide materials and services that meet your organization's specifications and regulatory expectations.
Importance of Supplier Compliance Audits
Supplier compliance audits are a regulatory requirement and critical business practice. Regulatory agencies expect manufacturers to exercise appropriate oversight of suppliers, and responsibility for material quality ultimately rests with the manufacturer. A robust supplier audit program protects product integrity and regulatory standing.
Key Benefits
Risk Mitigation: Identify quality and operational risks early, preventing recalls and production delays
Regulatory Compliance: Demonstrate compliance with FDA, EMA, and ISO supplier oversight requirements
Cost Reduction: Prevent incoming material defects and reduce rework and scrap
Supply Chain Visibility: Gain confidence in supplier capacity, capability, and stability
Product Integrity: Control material quality at the source to ensure final product safety and quality
Types of Supplier Compliance Audits
Different audit types serve specific purposes at different stages of the supplier relationship. Understanding these distinctions helps organizations develop a comprehensive supplier management strategy.
1. Qualification Audits
Conducted before approving a new supplier to assess their quality systems, capacity, and capability to meet requirements.
Timing: Pre-approval.
Scope: Comprehensive evaluation of all relevant areas.
2. Re-certification Audits
Periodic audits (typically every 2-3 years) of existing suppliers to verify continued compliance and system effectiveness.
Frequency: Based on risk assessment and performance history.
Scope: Full facility assessment or focused on specific areas.
3. Targeted/Cause Audits
Conducted in response to specific concerns such as quality issues, failed test results, or audit findings.
Timing: As needed based on risk.
Focus: Root cause of the specific issue.
4. Regulatory Follow-up Audits
Conducted after a supplier receives a regulatory inspection with findings to verify corrective action implementation.
Timing: Post-regulatory finding.
Purpose: Validate effectiveness of corrections.
5. Second-Party vs. Third-Party Audits
Second-party: Audits conducted by your organization directly at the supplier facility. Third-party: Audits conducted by independent external auditors or certification bodies. Both approaches are valid; many organizations use a combination based on risk and resources.
What to Include in a Supplier Compliance Audit
A comprehensive supplier compliance audit evaluates the supplier's ability to consistently deliver quality products and services while maintaining compliance with applicable regulations. Auditors assess operational systems, quality controls, documentation, and management commitment. The scope of the audit typically covers the supplier's manufacturing processes, quality systems, and documentation practices relevant to the materials or services they provide to your organization. Key Audit Areas:
Quality Management System: QMS documentation, responsibility assignments, management review, internal audits
Personnel & Training: Qualifications, training records, competency assessments, regulatory knowledge
Facilities & Equipment: Building conditions, equipment maintenance, calibration, environmental controls
Manufacturing Processes: Process documentation, validation, change control, process capability
Quality Control & Testing: Test method validation, analytical equipment, reference standards, data review
Raw Materials & Sub-suppliers: Supplier qualification, incoming material testing, traceability
Documentation & Records: SOPs, batch records, test results, complaint handling, record retention
Regulatory Compliance: Adherence to GMP/regulatory requirements, inspection history, corrective actions