What Is a GMP Compliance Audit?
A GMP Compliance Audit is a systematic, documented process used to evaluate whether a manufacturing operation complies with Good Manufacturing Practice (GMP) regulations and standards. These audits are essential for verifying that a company's processes, systems, and controls meet the stringent requirements set by regulatory bodies such as the FDA, EMA, and other health authorities.
How to Prepare for a GMP Compliance Audit?
Thorough preparation is important to a successful GMP compliance audit. Organizations should approach preparation as an ongoing process rather than a last-minute effort. Here are the steps you can take to be prepared for a GMP Compliance Audit:
Pre-Audit Preparation Steps
1. Conduct an Internal Assessment - Perform a comprehensive self-audit using GMP guidelines to identify gaps and deficiencies before the official audit. Document all findings and corrective actions.
2. Review Documentation - Organize and validate all required documents: SOPs, batch records, validation reports, training records, and maintenance logs. Ensure completeness and accuracy.
3. Update Standard Operating Procedures - Ensure all SOPs are current, reviewed, and approved. They should reflect actual manufacturing processes and be readily accessible to personnel.
4. Verify Personnel Training - Confirm that all personnel involved in manufacturing and quality have completed relevant GMP training. Maintain comprehensive training records with dates and topics covered.
5. Validate Equipment and Systems - Ensure all manufacturing equipment, analytical instruments, and computer systems are validated, calibrated, and maintained with documented evidence.
6. Correct Outstanding Issues - Address findings from previous audits and internal assessments. Document closure of corrective actions with evidence of effectiveness.
Facility and Environmental Preparation
Clean and organize manufacturing areas, storage facilities, and quality control labs
Verify proper environmental controls (temperature, humidity, cleanliness classifications)
Ensure proper segregation of materials and product areas
Check water systems, HVAC, and utilities for proper operation and maintenance
Team Preparation
Designate an audit coordinator and quality team members responsible for specific areas
Brief all relevant personnel on audit scope and their roles
Prepare department heads to answer technical questions about their operations
Ensure facility managers are available to escort auditors
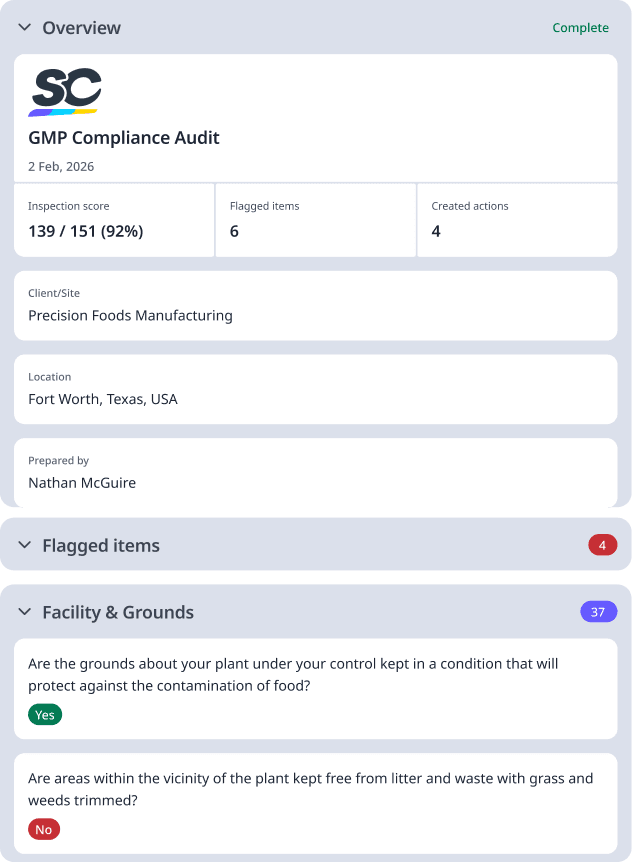
What to include in a GMP Compliance Audit?
A comprehensive GMP compliance audit should evaluate all critical aspects of manufacturing operations to ensure full regulatory compliance and product safety. Auditors will examine both your operational systems and the documentation that demonstrates compliance. The scope of the audit typically covers the entire product lifecycle from raw material procurement through manufacturing, quality control, and final product release:
Key Audit Areas:
Personnel & Training: Qualifications, training records, competency assessments, health programs
Facilities & Equipment: Building design, equipment validation, calibration, environmental monitoring
Raw Materials & Suppliers: Supplier qualification, incoming testing, traceability, rejections
Manufacturing Processes: Documentation, process validation, change management, deviation handling
Quality Control: Test validation, reference standards, data review, OOS investigations
Documentation & Records: SOPs, batch records, document control, data integrity
Quality Management System: Change control, CAPA, risk assessment, internal audits
Stability & Testing: Stability protocols, shelf-life data, finished product testing
Types of GMP Compliance Audits
Different types of audits serve specific purposes and are conducted at various frequencies. Understanding these distinctions helps organizations prepare appropriately and maintain continuous compliance.
1. Internal Audits (Self-Audits)
Conducted by your own quality team to identify and correct deficiencies before external audits.
Frequency: Annual minimum.
Benefit: Early detection of issues, continuous improvement, cost-effective.
2. Regulatory Audits (FDA/EMA Inspections)
Conducted by government agencies to verify compliance with applicable regulations. These are mandatory and typically unannounced.
Frequency: Every 2-5 years depending on risk and history.
Impact: May result in warning letters or enforcement actions if deficiencies are found.
3. Supplier Audits
Assess the compliance and capability of raw material suppliers and contract manufacturers.
Frequency: At qualification and every 2-3 years thereafter.
Purpose: Ensure supply chain quality and prevent material issues.
4. Third-Party Audits
Conducted by independent external consulting firms to provide objective assessment of GMP compliance. Often required by customers for certification.
Frequency: Typically annually.
Benefit: Independent evaluation and customer confidence.
5. Targeted/Focused Audits
Focus on specific areas or processes rather than full facility evaluation. Examples include environmental monitoring, data integrity, or cleaning validation audits.
Frequency: As needed based on risk or concerns.
6. Specialized Audits
Microbiological/environmental audits assess cleanroom classification and microbial control. Data integrity audits evaluate electronic systems and records (21 CFR Part 11).
Frequency: Ongoing or regular intervals depending on audit type.